In the first phase of the project, three showcase studies were successfully performed in model experiments under controlled conditions. They included experiments with Salmo trutta, Oreochromis sp. and Sparus aurata, representing the showcases from the temperate zone (fresh water), the tropics (fresh water) and the Mediterranean (marine) respectively. In addition, we were able to setup a robust analytical pipeline for the antibiotic compound (florfenicol), which is used as a model in our studies as it is used worldwide in aquaculture.
Our current analysis of the data clearly demonstrates that significant effects on the gut microbiome of the three fish species were visible, mainly during the phase of administration of the compounds under investigation. These manifested themselves in a clear reduction in diversity.
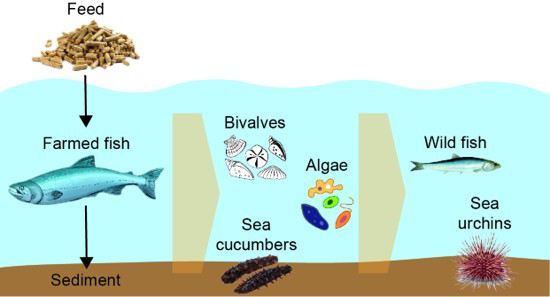
Effects became less significant when the application of the antibiotics was stopped, so much so that a regeneration of the microbiome was visible. But they still differed from the microbiome of the control group of fish, where no antibiotics had been applied, at the end of the experimental period. In addition, the application of compounds with antimicrobial/parasitic properties induced shifts in the microbiome of non-target organisms like mussels or seeweed. This may strongly affect the health and the fitness of these organisms and the subsequent food web structures in the affected ecosystems.
We also assessed functional consequences of these observed changes in the microbiomes of the administered fish as well as the non-target organisms. Here, we could identify a number of important genes which trigger host-microbe interactions which are affected by the administration of the antibiotic/parastic compounds tested both for target and non-target organisms.